
NuMega operates two 500 MHz Bruker NMR spectrometers, Avance II and AV-500. We have a variety of highly sensitive probes that can be interchangably fitted into our magnet. The diversity of both instruments enables us to carry out various experiments with a fast turnaround time. We perform the following experiments:
For H-1 NMR, there are four keys aspects you have to keep in mind while interpretting a spectrum:
Signals: The number of unique proton environments.
Integration: The area underneath the curve that corresponds to the ratio of hydrogens.
Chemical Shift: The best estimation of functionality based on shielding/ deshielding effects.
Splitting Pattern: The number of hydrogen neighbors which follows the N+1 rule unless a more complicated system is present.

Above are the relative chemical shifts for particular functional groups in an H-1 NMR spectrum. The more electronegative atoms near a proton, the more downfield or deshielded your signal will be. Unusual functionalities such as ring strain can alter expected chemical shifts. Tetramethylsilane (TMS) is a common reference standard with chemical shift at 0 ppm. Common chemicals shifts for NMR solvents include chloroform (7.26 ppm), methanol (3.34 ppm), acetone (2.17 ppm) and other common solvent shifts and impurities are linked within.
For C-13 NMR, there are a 2 key factors to keep in mind during interpretation:
Signals: The number of unique carbons in a given spectrum.
Chemical Shift: The position of a carbon in a spectrum based on shielding and deshielding effects.
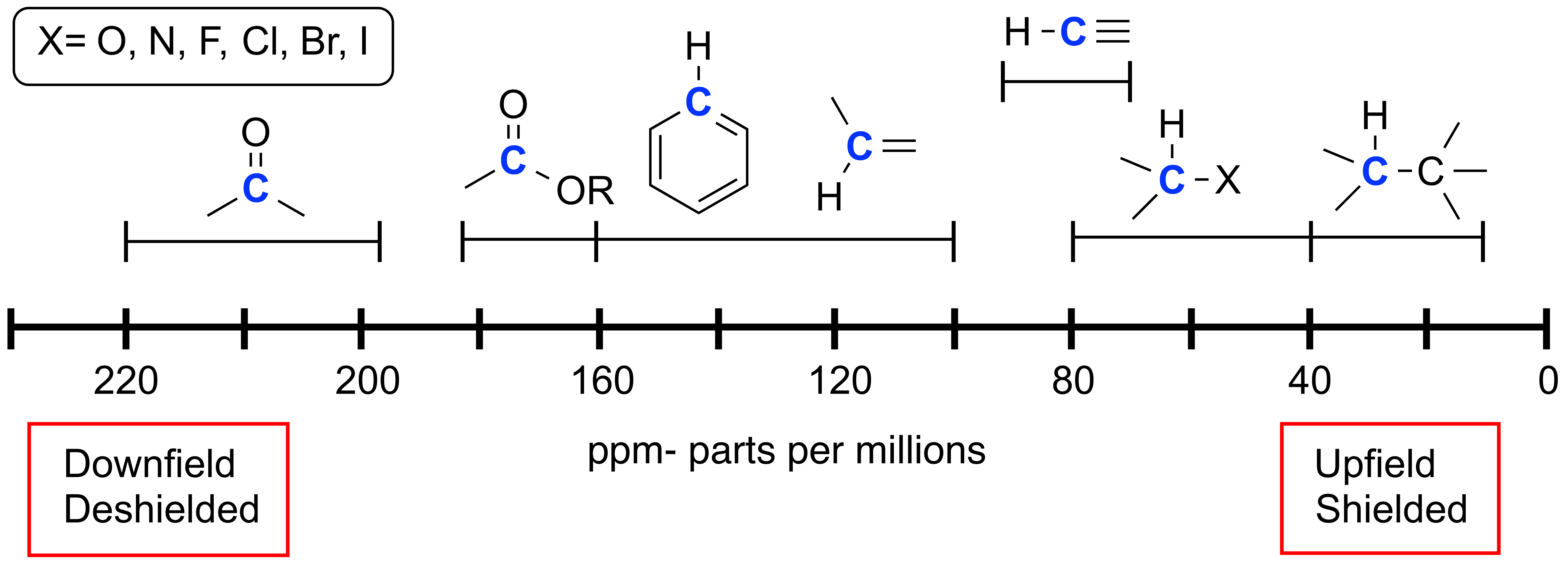
The more deshielded a carbon is, the higher the ppm value will be. While carbon NMR can be quantitative under certain test conditions normally you do not integrate carbon signals. Further experiments such as APT and DEPT experiments can give cues to how many attached hydrogens are on a particular carbon in the spectrum. Carbon NMR is usually ran as a proton-decoupled experiment which means that the carbon signals do not split. Although uncommon, this experiment can be ran as proton-coupled to show splitting patterns similar to that would be observed from a proton NMR.

Phosphorus NMR is a common technique for characterizing nucleotides or phospholipids since P-31 is in 100% relative abundances making this easier to measure in relation to other elements. Although less senstive than H-1 NMR, phosphorus NMR is a more sensitive technique than carbon NMR. Phosphorus NMR can be useful technique to determine purity since most phosphorus peaks show up in characteristic regions. Phosphorus NMR can be a key tool to differentiate mono-, di-, and tri- phosphoesters from each other due to characteristic chemical shifts. Common shifts for certain functional groups are shown above but signals can range from +250 to -250 ppm.

Fluorine NMR is another common nuclei detected for by NMR spectroscopy. F-19 is at 100% natural abundance and the aquistition of data is similar in peak intensity to what you would expect for running an H-1 NMR. Due to the electronics of fluorine, it is more sensitive and has a wider range of aquistition in comparison to H-1 NMR as seen above. This technique is common for the characterization of fluorine containing molecules.
Quantitative NMR is another common NMR technique. Quantitative NMR has multiple applications such as reaction monitoring, purity and stability studies to list a few. The theory behind quantitative NMR is by mixing a known quantity of a standard with a known quantity of your compound, there is an expected value of integration to compare the experimentally detected signal to. Due to the method, the standard must not have overlapping peaks with the sample NMR spectrum. Although this experiment is typically done with H-1 spectrums, the experiments can also be ran on C-13 spectra abeit a longer aquistion time.

NMR can be a useful tool in the characterization of polymers. You can use NMR to determine the relative ratio monomers and co-polymers that have been incorporated. It is important for polymer NMR to have a clear end group preferably one that does not overlap with peaks of your polymer unit as they are used in analysis for comparison to the monomer. With clearly defined end groups, you can use the NMR spectra to calculate the average molecular weight of your polymer through the ratios of end groups signals to that from the monomeric and copolymer signals. The stereochemistry of polypropylene groups can also be obtained from carbon NMR with the variations in stereochemistry through differences in chemical shift and number of signals. If all the methyl groups are oriented on the same side of polymer this is called isotactic. If the methyl groups alternate every other carbon which side they are oriented this is called syndiotactic. If the methyl groups stereochemistry is mixed with no defined pattern in orientation this is called atactic.
There are numerous Two-dimensional NMR (2D NMR) techniques that can give structural information about your molecule. Each experiment looks at the molecule in a slightly different scenario to give information about bond connectivity or spatial arrangement of a molecule. The data for 2D NMRs is plotted on two frequency axis with the central area showing correlation signals.
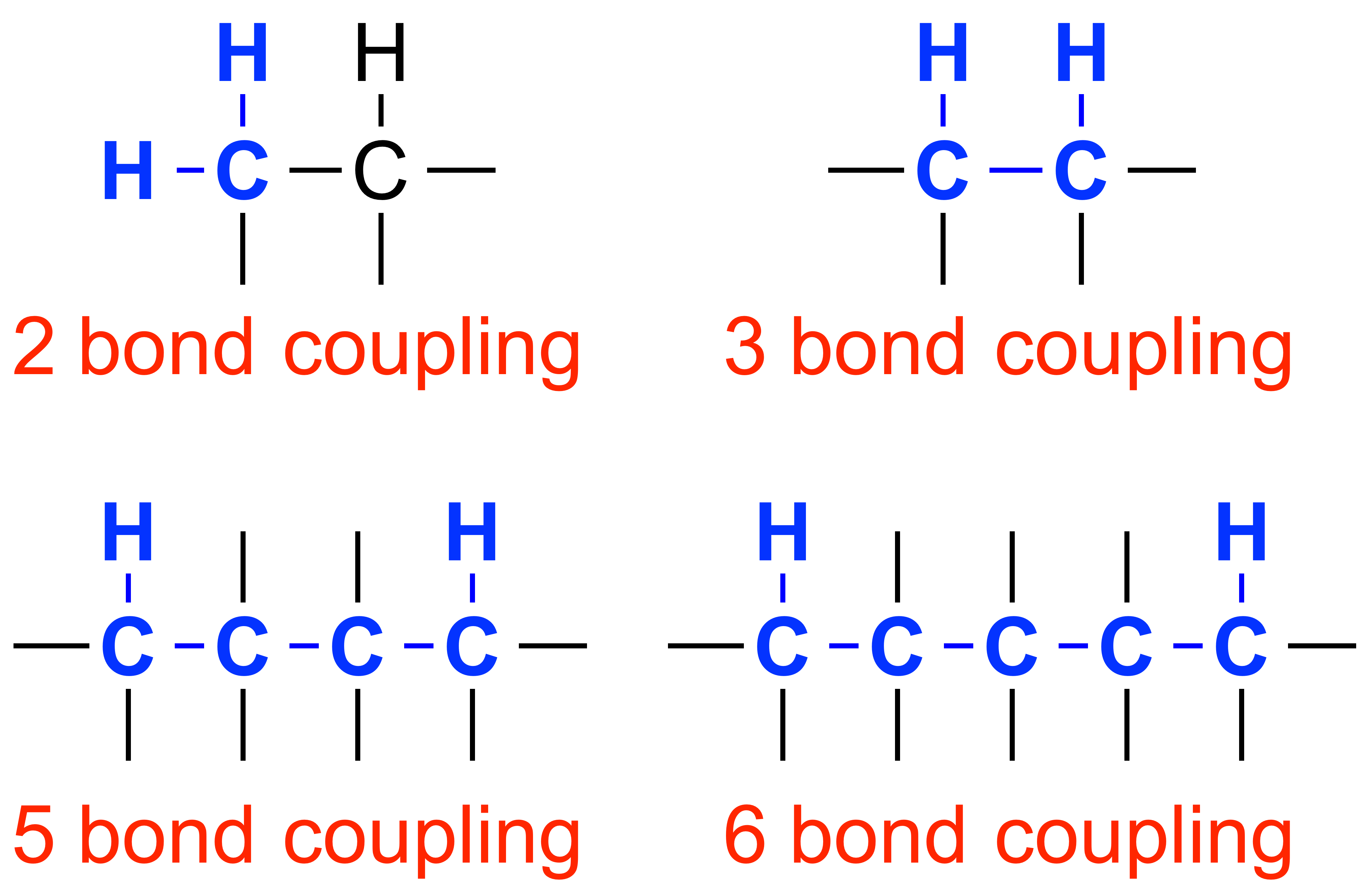
COSY: COrrelation SpectroscopY is a 2D spectroscopic technique used to view the connectivity of hydrogens through the bond. A cross peak arises when there is a correlation between signals on axis, indicating coupling between the hydrogens. COSY is used to view 2 to 3 bond couplings to assign proton environments for a molecule in a proton NMR. This technique is especially useful for differenitating multiple protons with similar functionality.
TOCSY: TOtal Correlation SpectroscopY, an experiment similar to COSY, can be useful in stereochemical assignment and to view 2 to 3 bond couplings, as well as couplings up to 5 to 6 bonds away. TOCSY especially useful for proton assignments on a ring.
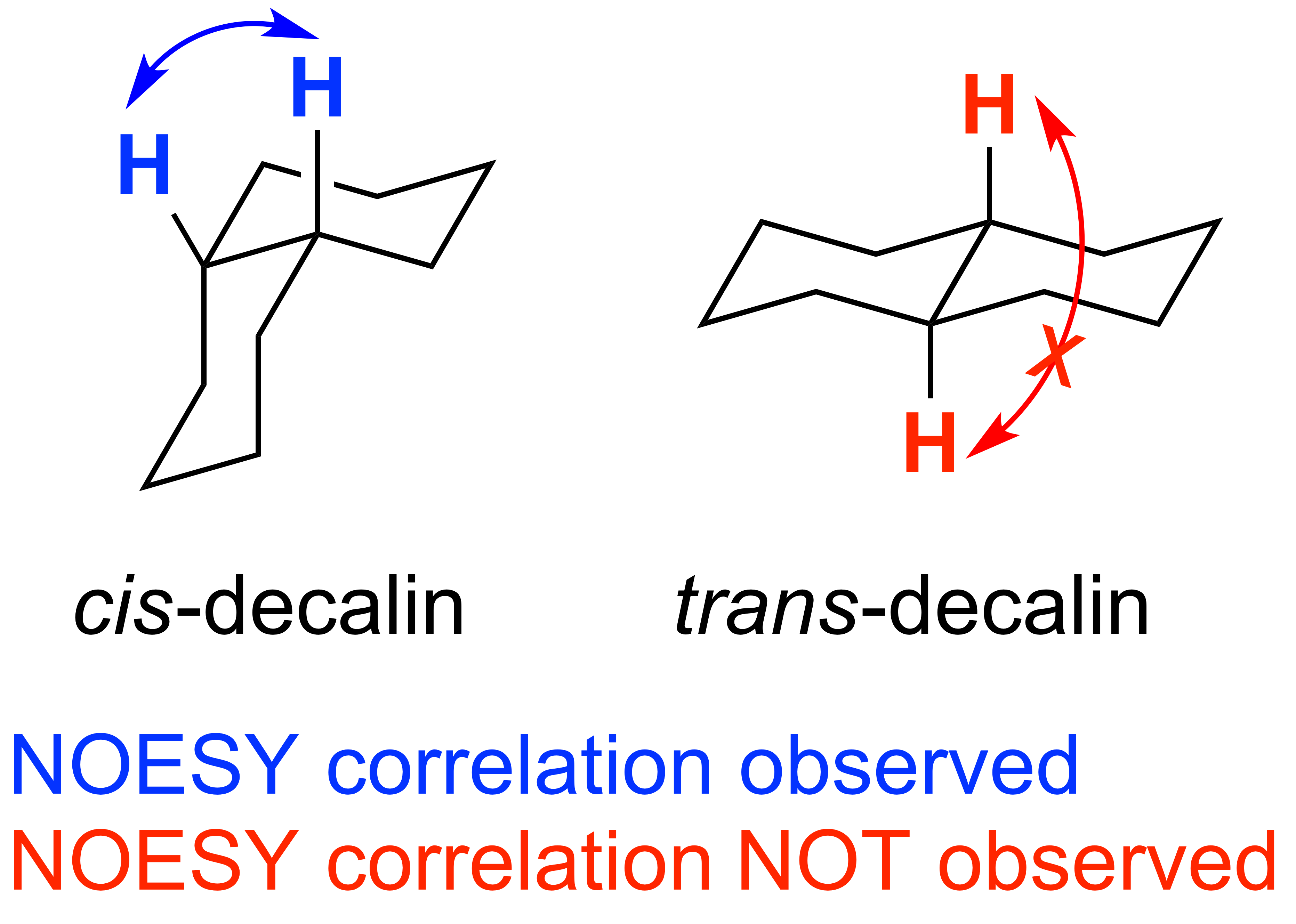
NOESY: Nuclear Overhauser Effect SpectroscopY is a spectroscopic technique used to view how hydrogen atoms are positioned in 3D space. Hydrogens will show a correlation if they are within 5 angstroms of each other. NOESY and ROESY are similar experiments, but NOESY is more appropriate on molecules with a low molecular weight (less than 1000 amu).
ROESY: Rotating Frame Overhauser Effect SpectroscopY, is another spectroscopic technique used to see hydrogens in 3D space but differs by shifting magnet equilbrium from the Z-axis to the X-axis. This difference in experimental parameters makes this a more appropriate for molecules with a high molecular weight (more than 1000 amu).

HMBC: Heternuclear Multiple-Bond Correlation spectroscopy, is a technique used to view hydrogen-carbon corrleations from 2-4 bonds. Direct 1 bond correlations are suppressed. HMBC is particular useful in assignments of conjugated systems.
HSQC: Heteronuclear Single-Quantum Correlation spectroscopy, similar to HMBC, is another technique used to view bond correlations between hydrogen and carbon. HSQC is particular useful in assignments of protons and carbon in a ring due to better sensitivity up to 5-6 bonds away.
A variety of NMR processing software exist for processing FID files for 1D or 2D NMRs including H-1, C-13, P-31 and other NMR experiments. A few common processing software are listed below.
MestReNova Mestrelabs developed MestreNova which is one of the easiest and commonly used NMR processing software. A free trial of the software is available before purchase.
TopSpin This is a software available from Bruker to process NMRs. This software can also be used to operate Bruker NMRs.
Delta This software was developed by Jeol and can process FID files as well as operate Jeol NMRs.
ACD/Labs This is another commonly used processing software for NMRS
NUTS This software was developed by Acorn and used to process NMRs
ChemDraw and ChemOffice are useful programs for drawing organic molecules and contains features like predict H-1 and C-13 NMR to assist with structural assignments of your molecules

NuMega operates a Perkin Elmer PE-SCIEX API-150 mass spectrometer equipped with an electrospray ionization source. Electrospray, developed in 1980s, is a mild ionization technique, which is useful for nonvolatile and unstable materials. Normally the parent ions are the dominant signals. Positive mode usually detects M+H+, M+Na+, and M+K+, while negative mode detects M+H-, and M+Cl-. Our detection range on the instrument is 30 – 3000 m/z. We report both the positive and negative ionization scans of your sample.
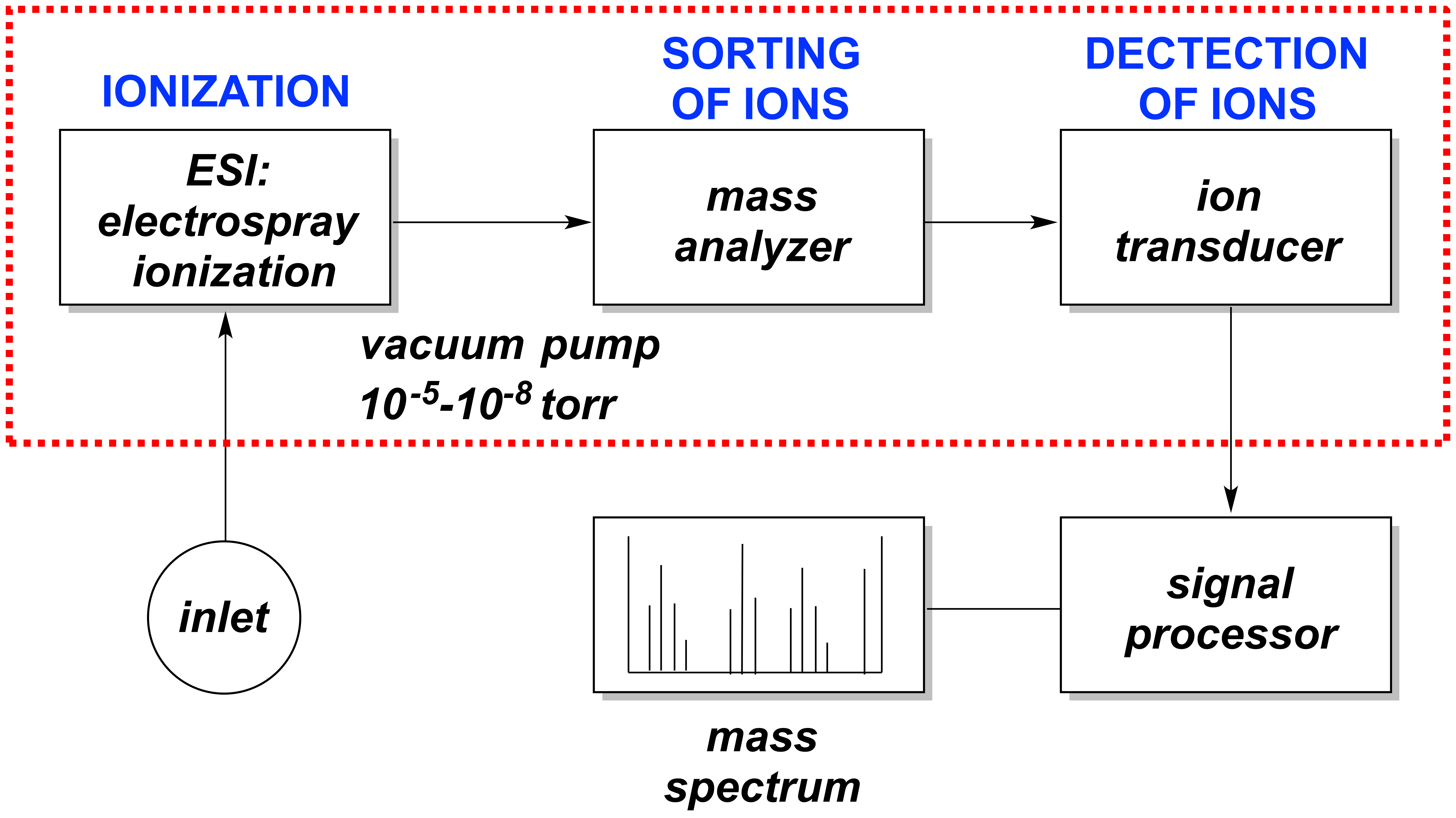
Mass Spectrometry (mass spec) is an analytic technique used to measure the mass of molecular ions. The mass spec operates by injecting a dilute sample of your molecule which then undergoes electrospray ionization (ESI) usually through protonation or cationization. The ions are dispersed in an electric field generating highly charged droplets from which solvent vaporizes. The resulting molecular ions are then introduced into the mass spectrometer. The ions are mass filtered by a quadrupole and are detected using a channeltron electron multiplier. The signal processor delivers the observed spectrum with the recorded mass-to-charge ratio.

NuMega operates a Perkin Elmer PE2400-Series II, CHNS/O analyzer for elemental analysis. The CHNS content is determined using the Pregl-Dumas method where your sample is prepared, introduced into the combustion chamber and burned at high temperature 925°F under pure oxygen. The resulting gas mixture and helium carrier gas pass through various reductive and catalytic zones to convert the gas mixture into CO2, H2O, N2, and SO2. The gasses are separated using gas chromatography and quantified with a thermal conductivity detector.
NuMega operates a Jasco P-2000 polarimeter for the measurement of optical activity in chiral molecules. Our current light source for the instrument is a sodium lamp with a 589 nm wavelength filter.The polarimeter operates by directing light through a polarized lens and through the sample cell to quantify how much the molecule in solution rotates the plane-polarized light. All samples that rotate to the right or dextrorotary are designated with a (+) for clockwise motion. All samples that rotate to the left or levorotatory are designated with a (-) for counter-clockwise motion. Our instrument is equipped with a temperature controlled chamber capable of maintaining temperatures anywhere from 15-40°C. We have 2 cell sizes: 3.5 x 100mm with a capacity for 3 mL of solvent and 10 x 100mm with a capacity for 10 mL solvent to accommodate the needs of your experiment. Our report includes the specific optical rotation, concentration, temperature, and path length of the cell.